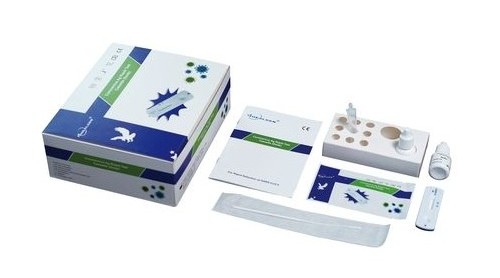
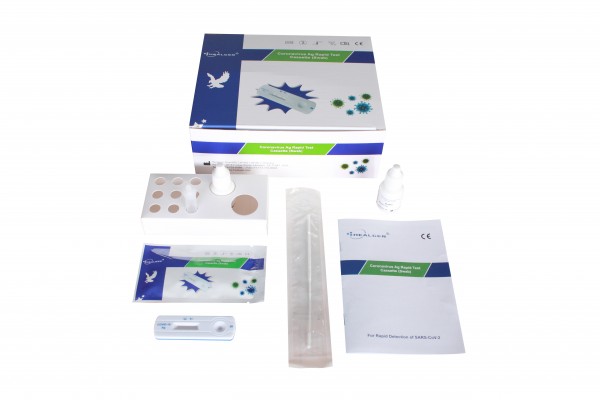










Healgen Coronavirus AG Rapid Test Cassette (CE Marked 15min Covid-19 Nasal Swab) x 20 Test Kit
We're always working hard to give our customers as much information on products and the best price possible. If you need any assistance or would like a quote please contact us and we will be happy to help
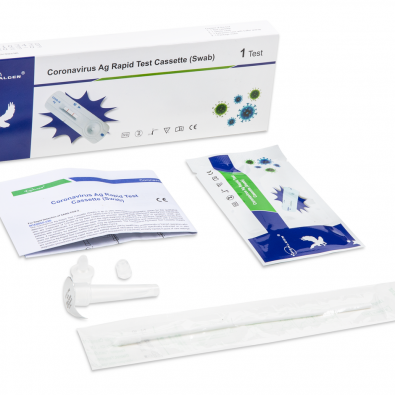
COVID-19 Rapid Antigen Test (CE Marked 15min Nasopharyngeal or Nasal Swab) 20 Test Kit (GCCOV-502a) is a rapid in vitro immunochromatographic lateral flow test assay for the qualitative detection of SARS-CoV-2 nucleocapsid protein antigen. Manufactured by Healgen for medical healthcare professional in vitro diagnostic use only.
For medical healthcare professional in vitro diagnostic use only. If you require testing for home use then please see our self tests HERE.
- Evaluated and Validated by the UK’s MHRA/DHSC: Approved test for use by schools, hospitals, and the army. Used by the NHS in the UK. More details HERE.
- French Government Validation: Full compliance with the Ministry of Solidarity and Health requirements for COVID-19 testing.
- German Government Validation: Full compliance with the Federal Institute for Drugs and Medical Devices COVID-19 testing requirements.
- Detects COVID-19 Variants including Omicron: Study by PHE and University of Oxford confirms variants, including Delta, are detectable. More details HERE.
- EU Approved Test: One of the few tests approved for use across all member states of the EU.
- Easy to Use: No special equipment needed, intuitive visual interpretation
- Rapid: Quick sampling with results in 15 min
- Convenient: All necessary reagents provided & no other equipment needed
- CE Marked: For professional use
- Accurate: 99.60% Specificity, 97.25% Sensitivity & 98.73% Accuracy
- ISO13485: Healgen manufacture in accordance with their ISO13485 Quality Management accreditation. They also hold many additional ISO accreditations.
Contents:
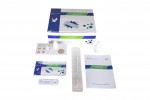
Each kit contains all the components required to run 20 tests:
- 20 Individually sealed test cassettes
- 20 Sterile swabs
- 20 Extraction Tubes and Tips
- 2 Extraction Buffer Vials
- 1 Workstation
- 1 Instruction manual (IFU)
Who can run these tests?
The test should be only be used in accordance with the instructions supplied and results interpreted by or under the supervision of a Medical Healthcare Professional. The test is not for home use. If you require testing for home use then please see our self tests HERE.
What does it do?
The novel coronaviruses belong to the β genus. COVID-19 is an acute respiratory infectious disease. People are generally susceptible. Currently, the patients infected by the novel coronavirus are the main source of infection; asymptomatic infected people can also be an infectious source. Based on the current epidemiological investigation, the incubation period is 1 to 14 days, mostly 3 to 7 days. The main manifestations include fever, fatigue, and dry cough. Nasal congestion, runny nose, sore throat, myalgia, and diarrhea are found in a few cases. This test is for the detection of SARS-CoV-2 nucleocapsid protein antigen. Antigen is generally detectable in upper respiratory specimens during the acute phase of infection. Rapid diagnosis of SARS-CoV-2 infection will help healthcare professionals to treat patients and control the disease more efficiently and effectively.
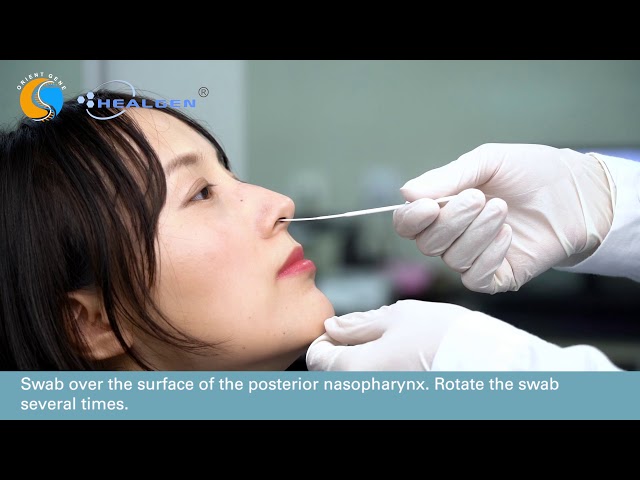
Healgen Coronavirus AG Rapid Test Cassette Video Tutorial
Video tutorial for performing the Healgen Coronavirus (Covid-19) Ag Rapid Test Cassette.
-
Does the test detect COVID-19 variants including Omicron?
-
Yes. The nucleocapsid phosphoprotein of SARS-CoV-2 was used as the main detected target in the Healgen Coronavirus Ag Rapid Test Cassettes (Swab) and Rapid COVID-19 Antigen Self-tests. This antigen test can therefore be used to detect both wild and mutant SARS-CoV-2 including the Alpha, Beta, Delta, Gamma, Lamda and Omicron Variants.
A statement from the manufacturer Helagen can be viewed HERE.
-
Who can run these tests?
-
What does the test do?
-
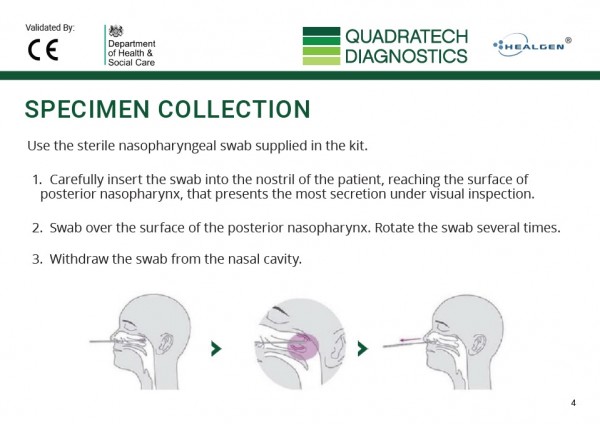
What samples are required to run the test?
-
What is the difference between Antigen testing and Antibody testing?
-
What is the testing time?
-
What are the possible results?
-
Is the test CE-marked?
-
Are these tests approved by the UK Medicines and Healthcare products Regulatory Agency (MHRA)?
-
Does this test meet any other Government standards?
-
Are these tests registered with the UK Medicines and Healthcare products Regulatory Agency (MHRA)?
-
Does the test need any additional components?
-
Who can buy these tests?
-
How do the Healgen and Innova tests compare?